 
Here is the table representing 118 elements of the periodic table. Such elements are present in Lanthanides and Actinides groups. f-block elements: These elements have the last electron filled in their f-subshell.d-block elements are also called transition elements as they have partially filled d-orbitals in their ground state. These elements have the last electron filled in their d-subshell. d-block elements : The elements present in groups 3 to 12 are d-block elements.The elements included in this block from different groups are termed as Boron Family (Group 13), Carbon Family (Group 14), Nitrogen Family (Group 15), Oxygen Family (Group 16), and Fluorine Family (Group 17). These elements have the last electron filled in their p-subshell. p-block elements : The elements included in groups 13 to 17 are p-block elements.The elements included in the s-block are Alkali metals and Alkaline Earth Metals. s-block elements: The first and the second group elements that have the last electron filled in the s-subshell are called s-block elements.Further, the elements are divided into different blocks. The 118 elements are arranged in 7 periods and 18 groups as shown below. ISRO CS Syllabus for Scientist/Engineer ExamĬlassification of Elements in the Periodic Table.ISRO CS Original Papers and Official Keys.GATE CS Original Papers and Official Keys.Full Stack Development with React & Node JS(Live).Java Programming - Beginner to Advanced. 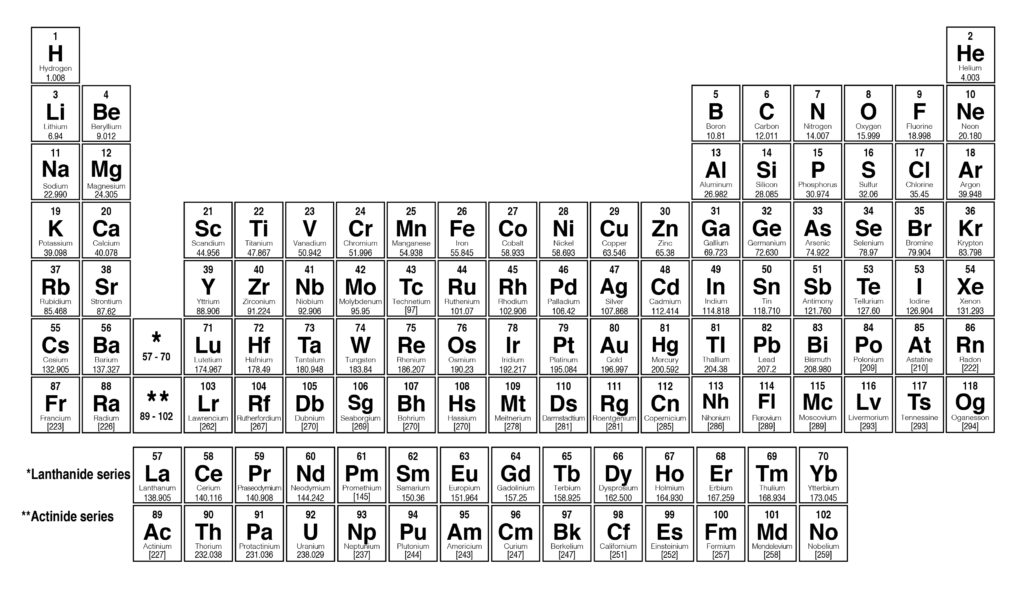
OS DBMS CN for SDE Interview Preparation.Data Structure & Algorithm-Self Paced(C++/JAVA).Full Stack Development with React & Node JS (Live).Data Structure & Algorithm Classes (Live). 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
